Brain Treatment Center Glenview offers FDA-approved TMS therapy for Major Depressive Disorder, covered by Medicare for eligible patients in the Chicago area. If antidepressants haven’t worked — or have been difficult to tolerate — TMS is a non-invasive, medication-free alternative with lasting results. Our team handles insurance verification from start to finish.
When Depression Doesn’t Improve With Medication
Depression is a serious medical condition that can affect energy, concentration, sleep, relationships, and overall well-being.
While antidepressant medications help many people, others may find them ineffective, difficult to tolerate due to side effects, or inconsistent with their personal treatment preferences.
If you feel stuck, you are not alone. And additional treatment options are available.

FDA-approved for Depression

Noninvasive Treatment

Medication-free

Medicare Accepted
Insurance Coverage for rTMS

Medicare covers FDA-approved rTMS for Major Depressive Disorder when medical necessity criteria are met. Brain Treatment Center Glenview, located in the northern Chicago suburbs, accepts Medicare for this treatment. You may qualify if:
- You have a diagnosis of Major Depressive Disorder
- Antidepressant medications have not provided sufficient improvement
- A physician determines that rTMS is medically appropriate
Our team will verify your benefits and guide you through the approval process.

TRICARE for Life also covers FDA-approved rTMS for Major Depressive Disorder when medical necessity criteria are met. If you are a Medicare-eligible beneficiary enrolled in TRICARE for Life, you may qualify under the same criteria above.
We will review your benefits and confirm eligibility before treatment begins.
What Happens Next?
- Submit the short form below
- We verify your insurance benefits (Medicare or TRICARE for Life)
- Schedule your consultation
- Begin treatment if approved
Clear. Direct. No pressure.
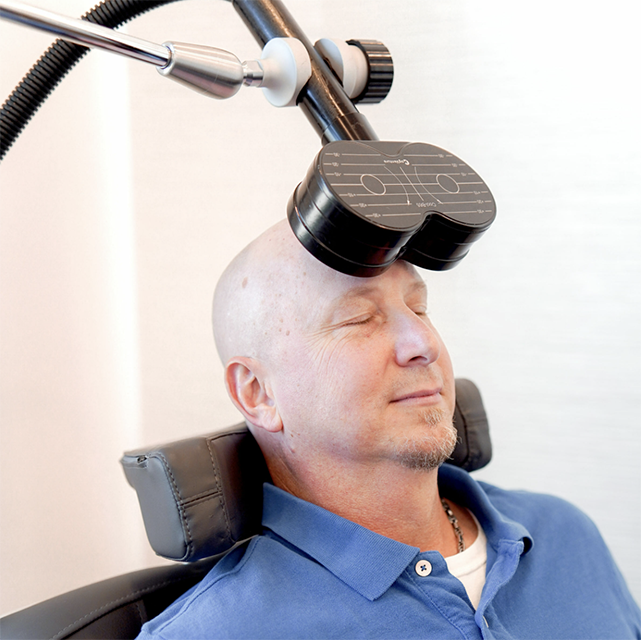
Check Your Eligibility
Complete this form, and our team will contact you to review your coverage and next steps. There is no obligation, and your data is secure.
Frequently Asked
Does insurance really cover rTMS?
Yes, Medicare and TRICARE for Life cover rTMS for Major Depressive Disorder when eligibility criteria are met.
Are there side effects?
rTMS is generally well tolerated, with mild scalp discomfort or headache, which dissipates, being the most common side effect.
Is rTMS safe?
Yes. rTMS is FDA-approved as a treatment for depression and has been safely used for over a decade.
Does it hurt?
It is painless. Most patients report a light tapping sensation on the scalp.
Does Medicare cover TMS therapy in Illinois?
Yes. Medicare covers FDA-approved TMS (also called rTMS) for Major Depressive Disorder in Illinois and nationwide. Coverage is available when medical necessity criteria are met, including prior antidepressant trials. Brain Treatment Center Glenview accepts Medicare and can verify your specific benefits before your first appointment.
How do I know if I qualify for Medicare-covered TMS?
Eligibility generally requires a diagnosis of Major Depressive Disorder and a documented history of antidepressant medication that did not provide adequate relief. A physician will review your case as part of your initial consultation. Our team will also verify your coverage directly with Medicare before treatment begins.
Where is the Brain Treatment Center located?
We are in Glenview, Illinois, serving patients throughout the North Shore and northwest Chicagoland area, including Northbrook, Evanston, Wilmette, Winnetka, and surrounding communities.

Physician-Led Depression Care in Glenview
Your evaluation and treatment are overseen by a licensed physician experienced in neuromodulation and depression care. Every patient receives a comprehensive medical assessment to determine whether rTMS is appropriate before treatment begins.
“After a month of treatment, I was grateful to be depression-free and medication-free for the first time in a very long time.”
—Patient after rTMS treatment
FDA-approved | Medicare & TRICARE for Life Accepted | Non-Invasive
If Depression Hasn’t Improved with Medication, Medicare-covered rTMS May Be Your Next Step.
Backed by Independent Clinical Research
Repetitive Transcranial Magnetic Stimulation (rTMS) is supported by a substantial and growing body of peer-reviewed research.
Multiple randomized controlled trials, meta-analyses, and large observational studies have shown significant reductions in depressive symptoms, meaningful remission rates, and strong safety outcomes, particularly in those who have not responded adequately to medication.
The therapy’s FDA clearance reflects this clinical evidence base, and ongoing research continues to refine treatment approaches for both civilian and military populations.
Selected Research Highlights
Adjunctive rTMS After Two Medication Failures (July 2023) – Findings showed clinically meaningful antidepressant effects, with evidence supporting remission in some patients when used alongside medication.
Psychiatric News (October 2022) – After reviewing a decade of outcome studies, researchers concluded that TMS should be considered alongside pharmacotherapy and psychotherapy as a first-line treatment for moderate to severe Major Depressive Disorder.
Meta-analysis of 65 Randomized Controlled Trials (October 2022) – Data from nearly 3,000 patients confirmed rTMS as effective in improving symptom reduction, response rates, and remission in depression.
Scoping Review of Treatment-Resistant Depression (June 2022) – 16 of 17 studies reviewed found rTMS to be effective, safe, and well tolerated in individuals with treatment-resistant depression.
Multisite Observational Study (2013) – Demonstrated significant improvements not only in depressive symptoms but also in quality of life and functional outcomes in routine clinical practice.
Stanford Accelerated TMS Study (2021) – A controlled trial reported remission in nearly 80% of participants using targeted, accelerated stimulation protocols.
Long-Term Evidence Reviews (2019–2022) – Consistently concluded that rTMS is an effective and safe treatment option for Major Depressive Disorder, including treatment-resistant cases.
Want to Know More?
Read more about TMS for Depression here…
